Part I: Motivation
In developing any new system for capturing carbon dioxide, we want to know how well the system is working. Cyan captures carbon dioxide through a reaction with powdered calcium hydroxide sorbent (input material) to produce calcium carbonate, as shown:
Ca(OH)2 + CO2 ----> CaCO3 + H2O
The output material from a classic Cyan box is a crumbly mixture of both solids, plus residual water. If we can determine the amount of CaCO3 that is in the output material, we can calculate the amount of captured CO2 using standard Chem 101 techniques.
A weighing technique is used with the original Cyan, as developed by Dahl Winters (https://www.openairforum.org/t/cyan-project-progress/105). It assumes the output consists of a mixture of CaCO3 with remaining unreacted Ca(OH)2 . From the increase in weight, the amount of captured CO2 can be computed. This method requires great care in weighing the input and output material; weight of the coffee filter must be accounted for; there must be no loss or spillage of the material; it is essential that the output material be completely dry before weighing. Unavoidable errors can come from impurities in the input material or the water used for moistening it. But with proper care, we can assume reasonable accuracy, and encouraging reports of the original Cyan’s effectiveness have been given.
With other potential Cyan architectures, however, it can be difficult to recover all of the output material, making an accurate result from the weighing method infeasible.
We would like to have an alternative measurement available as a confirmation of the results from the weighing method. The gold standard would be a careful quantitative analysis procedure in a chemistry lab, but few people have the necessary equipment and supplies.
I propose here a method where a sample of the output is weighed out and added to distilled water. Then the CaCO3, being insoluble, will sink to the bottom and the remaining Ca(OH)2 will dissolve (up to its solubility limit). Since Ca(OH)2 is a strong base and fully ionizes in solution, a measurement of the electrical conductivity (EC) of this solution with an appropriately calibrated sensor should make it possible to determine the concentration of Ca(OH)2 remaining in the sample. From this, we can find the proportion of CaCO3 in the output material, thence the amount of CO2 captured. This is similar to a method published here (Burns, 2005) and also used in my earlier Aqueous Cyan experiments.
Scientific-grade EC meters are expensive. I will show how one can build a sensor that is adequate for our purposes, consisting of only a processor such as an Arduino, and a few other inexpensive items. This new design improves on my earlier one. Further details of the method will follow in a later post.
Part II: Building a Conductivity Meter
In this post, I describe how to build an inexpensive meter to measure the electrical conductivity (EC) of a solution. The intent is to provide enough instructions so that you can build one, too.
For background, you might want to read the following articles:
The key point to note is that you can’t measure EC in an ionized solution with a conventional ohmmeter. The test probes, having a positive and a negative terminal, will cause the ions to drift, resulting in electrolysis and a change in the conductivity of the solution to be measured. One workaround is to put a pulsed voltage on one electrode, making the measurement quickly before turning off the electrode.
The tester that I built for Cyan is inspired by the “Three Dollar EC Meter” described here: Three Dollar EC - PPM Meter [Arduino]. I added a few refinements to improve the accuracy; however this scheme omits the temperature probe and compensation. It’s assumed that measurements will be done at a relatively constant room temperature. A similar meter was used in my earlier Aqueous Cyan experiments.
This tester requires a microcontroller with analog inputs. I used an Adafruit Feather M0 Basic, which contains a SAMD21 ARM M0 processor; an Arduino or similar would work just as well with modifications. Here is the test circuit:
My probe was built by simply soldering wires to a 2-pin through-hole header; heat-shrink tubing is important for insulation and strain relief. Many other designs are possible depending on what you have on hand. Here is a picture of the completed device.
Resistor R1 is a known value that can be modified according to measurement needs. R2 represents the unknown resistance that we want to measure between the electrodes of the probe. The outputs labelled V1 and V2 are alternately pulsed HIGH, and an analog measurement is taken on V1, V2, and Vout. The program leaves both outputs LOW for 10 msec between measurements. The value of R2, is calculated from

The final step is to compute the EC in units of microSiemens (uS) / cm, with
EC = k / R2
where the calibration constant k depends on the geometry of the individual probe and must be experimentally determined. The complete program is available on Github here:
To determine k, I mixed up a saturated solution of Ca(OH)2 in distilled water and adjusted k in the source code to make results match published data ( Burns, 2005, and others).
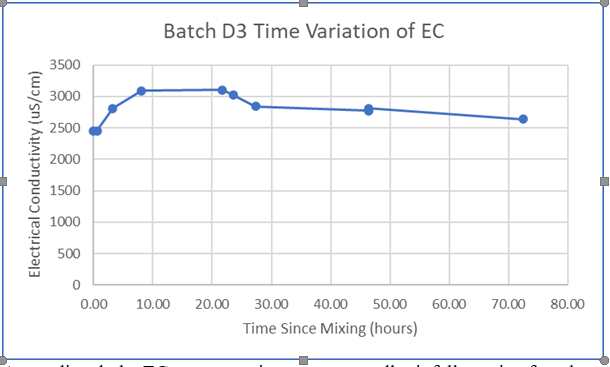
As a proof of concept, I mixed several solutions of Ca(OH)2 with varying concentrations and measured the EC with this setup. Results are shown here:
The pink line labelled “Saturation” indicates the maximum amount of Ca(OH)2 that can be dissolved at room temperature; any additional Ca(OH)2 added after this point will not dissolve, and results will level off at about 8000 uS/cm. The near-linear characteristic below saturation is a promising indication for this scheme.